Because the conditions that cause sudden adult death syndrome (SADS) can be inherited, it is important that if you are a blood relative in the immediate family of someone who has died of SADS you are evaluated for signs of these diseases.
There may also have been other sudden or suspicious deaths in your family, including cot deaths, suggesting that there may be an underlying inheritable condition. Below, we explain what is involved in the evaluation and describe the tests you may need to have.
Medical history
It is vital that a clear history of the deceased and his or her death is established, using the family’s and friends’ recollections as well as the reports of the coroner, pathologist, GP and police.
For example, fits brought on by exercise can be due to an underlying channelopathy such as long QT syndrome (LQTS) or catecholaminergic polymorphic ventricular tachycardia (CPVT), or a sudden cardiac death during sleep may have been caused by sodium channel LQTS or Brugada syndrome.
It is important to find out about any medications and any potentially dangerous drugs that the person may have taken before they died.
Your doctor may ask you if you have ever had symptoms such as blackouts or palpitations as these may suggest underlying heart disease.
Medical examination
A medical examination may help to discover if there is an inheritable structural heart disease in the family. For example, if there is mitral valve prolapse with leakage from the valve this will cause a “murmur” that a doctor can hear through a stethoscope.
Your doctor may suggest that you have some of the tests we describe below.
Tests marked with a * are non-invasive. “Non-invasive” means that it does not involve penetrating the skin or body
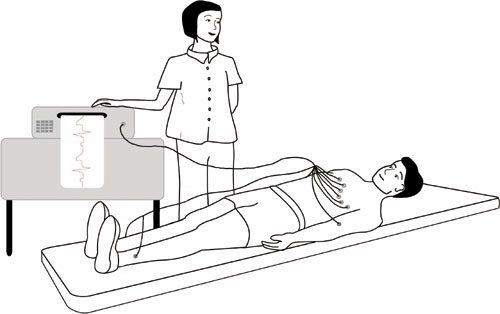
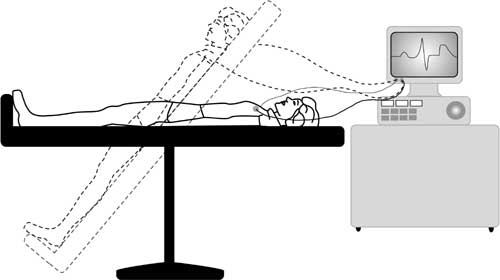
Electrocardiogram (ECG) *
This is the most basic test. It involves taping electrical leads onto your legs, arms and chest to take readings of the electrical activity of your heart. These are printed out onto a piece of paper for the doctor to examine.
The ECG involves lying down quietly and only takes 2-3 minutes to perform.
Signal averaged ECG *
This is an ECG that adds together the electrical readings from at least 250 heartbeats so that any very subtle variations can be seen – for example if the electrical impulses in the heart are being conducted more slowly. It is useful for diagnosing Brugada syndrome, progressive cardiac conduction defect (PCCD) or arrythmogenic right ventricular cardiomyopathy (ARVC).
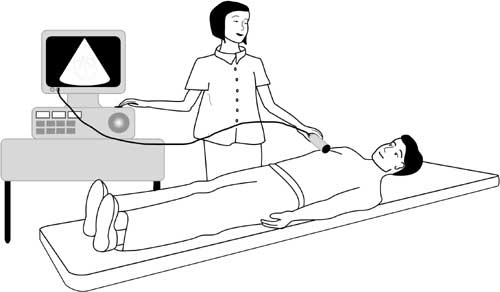
Echocardiogram * Also called an “ECHO”
This test uses ultrasound waves to look at the structure of the heart. It is useful for people whose ECG shows changes that could be caused either by a channelopathy or by uninherited heart disease that has damaged the heart – for example a previous heart attack that you may not have even been aware of.
An echocardiogram can also detect inheritable conditions such as cardiomyopathies and mitral valve prolapse.
The operator puts some clear gel on your chest and then places an ultrasound probe on it. The probe sends ultrasound beams into your body and their reflections are detected and used to generate images of the heart.
You can see different parts of your heart on a screen as the probe is moved around on your chest.
The test is similar to the ultrasound scan that is used to examine a pregnant woman’s unborn baby. It is completely painless.
Exercise test * Also called an “exercise ECG”
This test is an ECG but is recorded before, during and after a period of time spent exercising on a treadmill or an exercise bike. This allows the doctor to examine any changes in the electrical patterns that occur with exercise, and analyse any abnormalities. This test is particularly useful in detecting some of the features that are characteristic LQTS or CPVT.
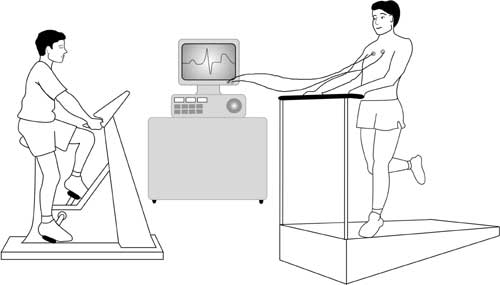
Cardiopulmonary exercise test (CPET/CPEX) *
Some hospitals may also ask you to do a cardiopulmonary exercise test. This test analyses the efficiency of the heart muscle by measuring the amounts of oxygen your body uses during exercise.
You will be asked to breathe into special equipment while you are exercising. If the efficiency of your heart is low, this may suggest that you have a cardiomyopathy (causing inefficient pumping of the heart).
Electrical leads from the ECG machine are taped to your body and you are monitored while you exercise either on an exercise bike or treadmill.
If you are having a cardiopulmonary exercise test, your doctor will ask you to breathe in and out of a special piece of equipment while you are doing the exercise, in order to monitor how efficiently your body uses oxygen.
Holter *
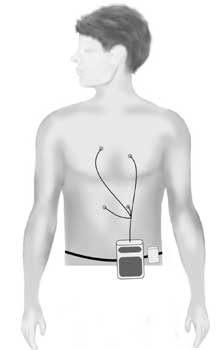
The Holter is a recording device that comes in two different forms:
• a small portable tape recorder (like a walkman), or
• a small digital device the shape of a pager.
You wear the device on a belt round your waist. Four or six ECG leads from the device are taped to your chest. The device records the electrical activity of your heart for 24 to 48 hours, or for up to seven days if a digital one is used. The doctor can then analyse the electrical activity and rhythm of your heart to find out if you have any arrhythmias (for example, the arrhythmias typical of LQTS and CPVT), or other features characteristic of LQTS.
Cardiomemo and event recorder *
These are more sophisticated versions of the basic Holter. Whenever you have an attack of symptoms, you can activate the device to record your heart’s rhythm. (You can also do this with the digital Holter.) The advantage of the cardiomemo is that it doesn’t have any leads, so you can just place it on your chest when you get symptoms, without having to put any leads in position.
Reveal© device
When it is difficult to assess or record a symptom because it only happens infrequently – as with blackouts – a Reveal© device can be used. The device, which is the size of a packet of chewing gum, is placed under the skin at the left shoulder. You will need to go into hospital as a day case to have this done. A small cut about 2cm long (just under one inch) is made and the device is inserted. The device monitors the heart’s rhythm and can record any abnormal events that it is programmed to detect. If anything happens, a small box with a button can also be placed on the surface of the skin over the Reveal© device. The Reveal may then be activated by pressing the button, causing the device to record the preceding 15 minutes of the heart’s activity. The device can then be “interrogated” by a computer at the hospital and the doctor can examine the recording. The device has a battery that can last up to two years if necessary.
Provocation tests (Ajmaline, flecainide and adenosine tests)
You may be asked to have this test if your doctor suspects Brugada syndrome. While you are having an ECG test you will be given an injection of ajmaline or flecainide (antiarrhythmic drugs). The test may show changes on the ECG that are typical of one of the channelopathies.
A fine plastic tube is inserted into a vein at the front of your elbow. The drug is injected over a short period of time (5-10 minutes) and you will be monitored for 20 minutes or a few hours afterwards, depending on the drug used. There is, however, a risk in 1 in 200 Brugada syndrome carriers or their immediate blood relatives of causing a potentially life-threatening arrhythmia during the injection. The test is always performed with appropriate facilities to protect patients from this risk.
Ajmaline is preferable as it lasts a shorter period of time in the circulation.
Adenosine (another short-acting chemical) is given under the same circumstances if Wolff-Parkinson-White syndrome (WPW) is considered a possible diagnosis.
Cardiac magnetic resonance (CMR) scan * Also called a “cardiac MRI”
This is a special kind of scan used to examine the structure of the heart and the nature of its muscle. It uses a magnetic resonance scanner that creates intense fluctuating magnetic fields around your body while you are inside the scanner. This generates the signals that make up the pictures produced. It is very useful for detecting the presence of fat and scarring in the heart muscle that is associated with ARVC.
Other tests
Coronary angiography and electrophysiological study (EPS)
Depending on the results of the above tests, your doctor may suggest that you have other tests such as coronary angiography or an electrophysiological study (EPS). Both these tests are performed in an X-ray laboratory that allows the body and any medical tools (such as cardiac catheter tubes or pacing wires) to be seen using an X-ray camera. You will be asked to lie down on a special moving table and will be given a local anaesthetic in your groin or wrist. The doctor will then place fine tubes, called cardiac catheters or electrodes, into blood vessels in your groin or wrist. These are gently passed through to the heart.
During coronary angiography the coronary arteries (the arteries that supply blood to the heart muscle) are injected with a dye to reveal any furring or blockages – coronary artery disease. (The ECG changes that are characteristic of Brugada syndrome or LQTS can sometimes be caused by coronary artery disease.)
An EPS (electrophysiological study) involves placing electrical leads inside the heart to analyse its electrical properties and induce arrhythmias. It may be useful in diagnosing WPW and PCCD and deciding on what treatment to give people with Brugada syndrome. If the extra pathway seen in WPW is detected at EPS it can be treated there and then by “burning” it away using high frequency radio waves. This procedure is called “RF ablation”.
There are other tests that may be used to provoke ECG features in LQTS such as “cold pressor tests”. A stimulus such as placing your hands in ice-cold water can bring out the ECG features of the condition. This does not appear to increase significantly the likelihood of making a diagnosis but is still used at some centres.
Tilt-table testing
Tilt-table testing is used to identify other common conditions that can cause blackouts – such as vasovagal syndrome or simple fainting – that tend to particularly affect young women and girls but have a very low risk of causing sudden death. These symptoms are very similar to the symptoms of more rare and potentially life-threatening conditions like channelopathies, so it is important to discover the cause of the blackouts so that the doctor can give appropriate treatment.
While you lie flat on a table, your blood pressure, pulse and ECG are monitored. The table is then tilted to an angle of 60 to 75 degrees and monitoring is continued. If nothing happens, a spray of a substance called glyceryl trinitrate (GTN) is given under your tongue as a stimulus and you will be monitored for another 10-15 minutes.
The table will then be returned to the flat position and the leads disconnected. The whole test takes around 45 minutes. If your blood pressure falls at the same time as you suffer your usual symptoms, this means that you have Vasovagal syndrome or a related condition.
Genetic testing
In most of the inherited conditions known to cause SADS, mutations of specific genes have been detected and are thought to cause a specific disease. So, in principle, if we could identify these mutations, we would be able to make a diagnosis in any DNA sample including any obtained from SADS victims at their autopsy or from their relatives who have given blood. Unfortunately this cannot be done at the moment because we don’t have complete knowledge of all the genes involved in any condition.
For example, only 6 in every 10 people known to have LQTS have mutations of known identified genes. Also, many variations in the DNA code are found in a large number of people and do not necessarily cause any disease. Most families with LQTS have mutations specific to them (“private” mutations) which can also make it difficult to decide whether it is the mutation that is causing the disease or not. As research progresses, more genes will be identified and there will be better tools to decide whether the impact of a mutation causes a disease.
